Greenhouse disease update for Downy mildew
Editor’s note: This article is from the archives of the MSU Crop Advisory Team Alerts. Check the label of any pesticide referenced to ensure your use is included.
Downy mildew is a water mold and related to the well-known Pythium and Phytophthora pests.Impatiens, rose, snapdragon, alyssum, pansy and salvia are susceptible to downy mildew and often suffer from the disease. Other crops such as cineraria, lisianthus, ranunculus, coleus and anemone may also become infected by a downy mildew pathogen although the disease problems are less frequent.
Snapdragons, roses, impatiens and other crops susceptible to downy mildew each have a unique downy mildew pathogen. While the disease may look similar on the different crops, they are quite different. The downy mildew on snapdragons cannot blight roses, nor can the downy mildew on impatiens spread to coleus.
Don’t forget to look under the leaves
Downy mildew may be restricted within the plant without causing severe injury. For instance, leaf spots are called “local infections” and are considered mild symptoms because the downy mildew is limited within the plant. However, if downy mildew advances and invades the internal or vascular part of the plant, the disease becomes “systemic” and is more destructive. When the infection is systemic, it affects the internal mechanism of the plant and hampers its ability to grow. Plants become stunted and yellowed with distorted leaves. When young plants and seedlings are infected, they generally do not survive. Downy mildew produces purplish, gray fuzz on the underside of the leaves. This can go unnoticed because growers are not accustomed to turning over the leaves to look for plant problems. Other than an especially severe case of Botrytis, no other disease causes gray or purplish fuzz on the underside of the leaf.
Look for the disease
Downy mildew is often explosive and early detection is critical. The downy mildew pathogen produces plenty of spores called sporangia that are spread via air currents to nearby healthy plants. Turn the leaves over to look for gray or purplish fuzz. Susceptible crops should be scouted weekly. Don’t forget to check the susceptible crops when you receive them and continue to check them once they’re potted especially if they’re in baskets that are hard to monitor.
Keep the production area clean
If you had downy mildew last year, this pest may persist in soil and growing media and mats via a thick-walled spore. The downy mildew pest can also reside in compost or cull piles where volunteer plants are likely.Once these volunteer plants become infected, spore production ramps up and the disease is off and running.
Downy mildew likes water
Limit this disease by venting, reducing the time that leaves are wet, and keeping relative humidity to a minimum. Water plants at a time of day that allows the foliage to dry quickly. Downy mildew is a water mold because it needs water to grow and develop. Any strategy that reduces water, leaf wetness, and relative humidity in a greenhouse will help limit downy mildew.
Apply effective fungicides preventively
Fungicides should be used for plants prone to downy mildew when the weather is favorable for an outbreak but before disease develops. A protectant fungicide such as mancozeb acts as a barrier to the downy mildew pathogen but is not absorbed by the plant so the spray program must begin prior to a disease outbreak. At MSU, downy mildew studies with snapdragons and vegetable crops have shown that weekly sprays of mancozeb effectively limit downy mildew if applications are made early and repeated frequently. Mancozeb sprays can serve as a relatively low cost, effective foundation for downy mildew protection. Systemic fungicides can be added to the fungicide program as needed. Systemic fungicides can be especially helpful in managing downy mildew because these products are absorbed by the plant in a limited way and can fight the pathogen from the inside out. Systemic fungicides include mefenoxam (example: Subdue MAXX) which is labeled as a soil drench only.Mefenoxam 2 applied to the soil performed well in most of our downy mildew trials with snapdragon. Stature, Adorn and Fenstop are also downy mildew fungicides.Cyazofamid is currently an unregistered product that provided exceptional control in two trials. Other products that appeared helpful but did not always perform as well as Stature include Compass, zoxamide/mancozeb (not registered), Echo, Aliette, and cymoxanil (not registered).
Downy mildew has a tendency to change and become resistant to the systemic fungicides (Subdue MAXX is an example).Since Subdue MAXX is especially at risk, it should only be used once in a production program and must be applied as a drench only.Using mancozeb is not likely to cause the pathogen to change and become resistant so using multiple applications of this fungicide is considered okay.Fungicides such as the strobilurins (Compass is an example) and Aliette should be used only in rotation with other fungicides that have a different mode of action to ensure that they remain effective and reliable tools.
Evaluation of dosages and sprays or drenches of fungicides for control of downy mildew of coleus, 2009
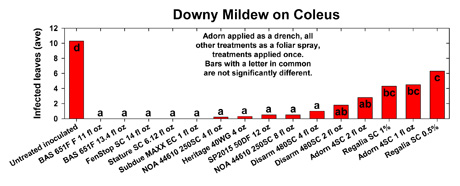
Disease pressure was severe in this trial with the untreated inoculated plants averaging 10.3 infected leaves.Although all treatments limited infection compared to the untreated control, differences were observed between the products tested.BAS 651F (both rates), FenStop SC, Stature SC, and Subdue MAXX EC were the only treatments that completely prevented infection in this trial.Although Adorn SC (both rates), applied as a drench, and the biological fungicide Regalia SC (both rates) limited infection compared to the untreated control,the number of infected leaves were still at levels unacceptable to growers.Although not statistically different, a rate response trend was observed with the high rates of Adorn SC, Disarm SC, and Regalia SC reducing infection compared to the lower rates.Phytotoxicity was not observed on any of the treated plants in this trial.



Acknowledgements
This research was funded in part by Floriculture Nursery and Research Initiative of the Agricultural Research Service under Cooperative Agreement #59-1907-5-553 and by the American Floral Endowment.
Dr. Hausbeck's work is funded in part by MSU's AgBioResearch.



 Print
Print Email
Email


